Krios session help guide
Getting ready for your Krios session
You have been assigned a Krios session at NCCAT. What are the next steps?
Schedule a pre-award meeting with the NCCAT User Office if your laboratory has not used the national center system before.
Make sure you have completed the pre-session forms:
[User registration][Shipping manifest][SEMC grid drop–off form]Review our shipping guide [here].
Verify the spokesperson of the proposal can login with your account at nccatweb.nysbc.org .
Register an account with Globus.org so you may download your data. See our quick guide or Globus’ FAQ.
Ship your grids to us
NCCAT User Office or your operator may reach out to you to go over your drop-off form.
- This help guide (Drop–off sheet addendum) outlines the process for capturing the necessary images for the updated online form.
A standard setting for one of our Gatan GIF/K3 systems would be:
Titan Krios GIF/K3 configuration:
Exposure Settings: 1.6-2.0 s, 40 ms frames (40-50 frames)
e Flux: ~20 e-/px/s with ~45-58 e-/Å total dose
Pixel Size: ~0.83Å/px (TIFF LZW format in counting or bin2 with an image dimension of 5760×4092 px)
Energy Filter: 20 eV slit
Defocus Range: -0.6 µm to -2 µm (nominal)
If you would like modifications let the NCCAT User Office know and update the Grid Drop Off Form ahead of your session.
A standard setting for our Selectris/Falcon4i system would be:
Camera: TFS Selectris/Falcon 4i, image shift
Exposure Settings: 6s [images saved in EER format]
e Flux: ~4 e-/px/s with ~47.69 e-/Å total dose
Pixel Size: ~0.73Å/px (EER format with an image dimension of 4096×4096 px)
Energy Filter: 10-20 eV
Defocus Range: -0.6 µm to -2 µm (nominal)
If you would like modifications let the NCCAT User Office know and update the Grid Drop Off Form ahead of your session.
Grid box numbering
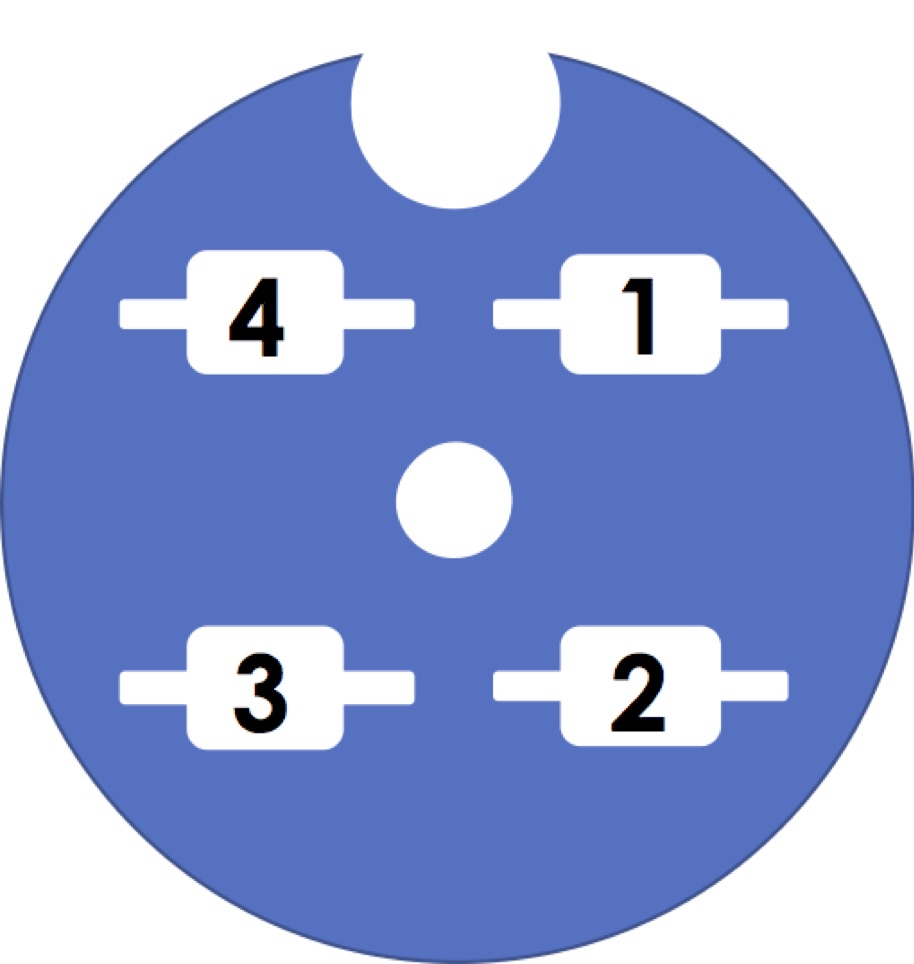
Users normally send us 1-2 buttons/boxes. Please try to have the grids as data collection ready as possible since our goal is to shift over to data collection as soon as possible. 2 users will share the cassette and we have a particular numbering system.
By default we will start with box1 grid1, then box1 grid2, box1 grid3 and box1 grid4. If more than one box is loaded then we will go to box2 grid1 and so forth.
An autoloader holds up to 12 cartridges. The maximum number of grids that will go into a cassette is 11 because in position 1 we keep a cross-grating grid for alignments and calibrations.
Your data collection session
Closer to your session day the NCCAT User Office will send an email with more details about your session. If you are accessing NCCAT under queued Krios access there may be modifications to your start date or time. Also, a primary operator will be assigned. Standard sessions are remote with a staff member setting up the microscope and assisting you with creating a data collection strategy.
Loading your samples into the autoloader. You may be sharing the cassette with another user.
Getting the microscope ready for data collection.
Determining a data collection strategy.
Communication: If you have any questions please let us know how we can assist. If email threads are becoming too long, then we recommend scheduling a video conference with your microscope operator.
How long is a Krios session?
- A typical session is 2-2.5 days, but may vary depending on the experiment and your access category.
How many grids can you collect during a session?
- We recommend planning an experiment that equates to on average 1 grid that our operator will queue up targets on per data collection day.
How many micrographs can you collect per session, day, etc…?
- Data collection rates will vary depending on the sample and grid. During our end of year checkouts we can reach 10,000 movies/day on an ideal sample (an apoferritin test sample on a gold/gold grid using multishot data collection using our standard settings). On user grids datasets can vary from 1,000 to 8,000 movies a day so the answer is not straightforward. You can arrange a pre-session meeting with your microscope operator to work out a more comprehensive experimental plan.
NCCAT uses Leginon for data collection.
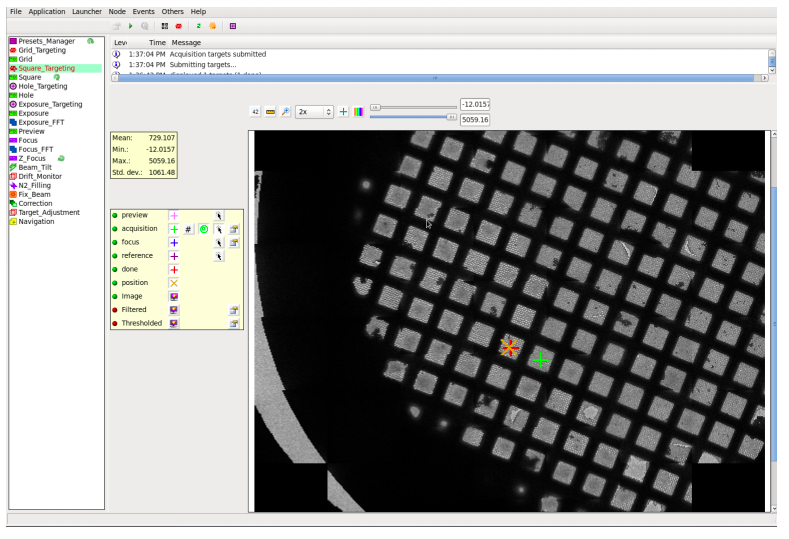
Leginon and Appion webtools
nccatweb.nysbc.org
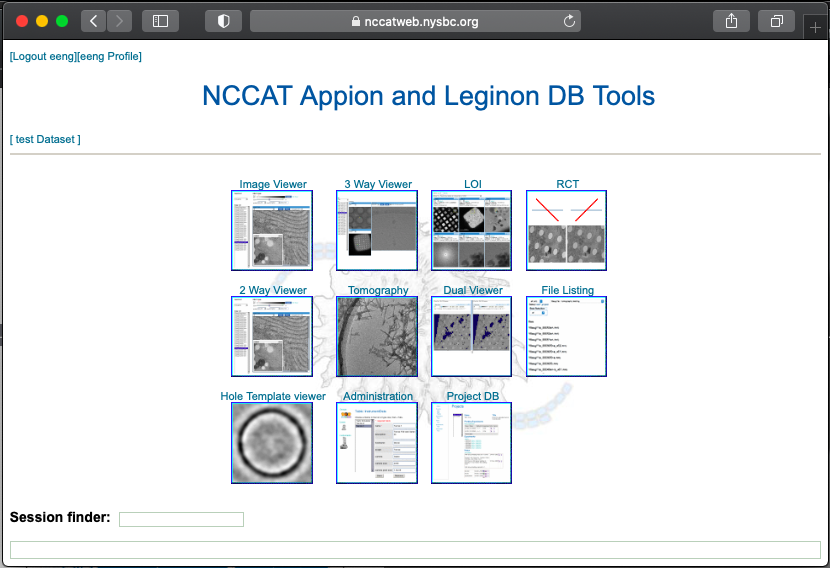
Given most of our Krios sessions are remote, for responsive feedback Leginon has webtools so the user may monitor the data collection and be able to advise our operators how to optimize their data collection strategy.
By default, we collect data under the spokesperson’s account. If additional accounts are required, these requests may be made to the NCCAT User Office during your pre-session meeting, or email correspondences, ahead of a scheduled data collection session.
If you have trouble with your accounts you may email the NCCAT User Office.
Image viewer
The main way to monitor your data collection is through the image viewer through our web interface.
Please remember to use your NCCAT proposal session assignment email as your main correspondence thread including the NCCAT User Office so we can promptly see to your queries. As part of this process, it is critical that you include your NCCAT proposal ID in the subject of all emails. If you would like to set up a video conference to better strategize on a data collection strategy let the NCCAT User Office know.
Note that NCCAT’s business hours are 9am-5pm ET during business days. Email correspondences outside this time may be delayed.
3-way viewer
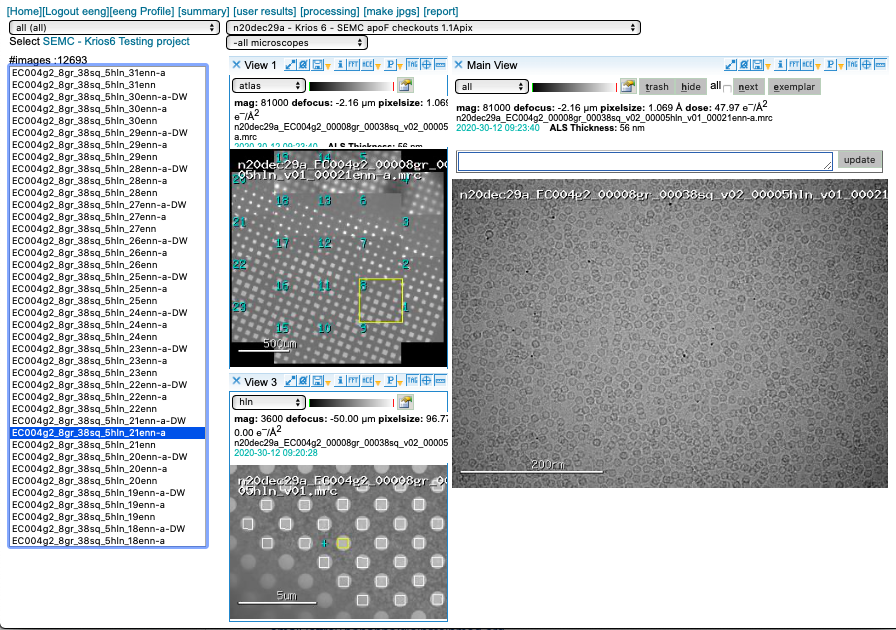
Appion pre-processing
Appion pre-processing has a few useful features. Although we prioritize data collection (acquisition of movies) we strive to provide as much feedback as possible.
Motion Correction
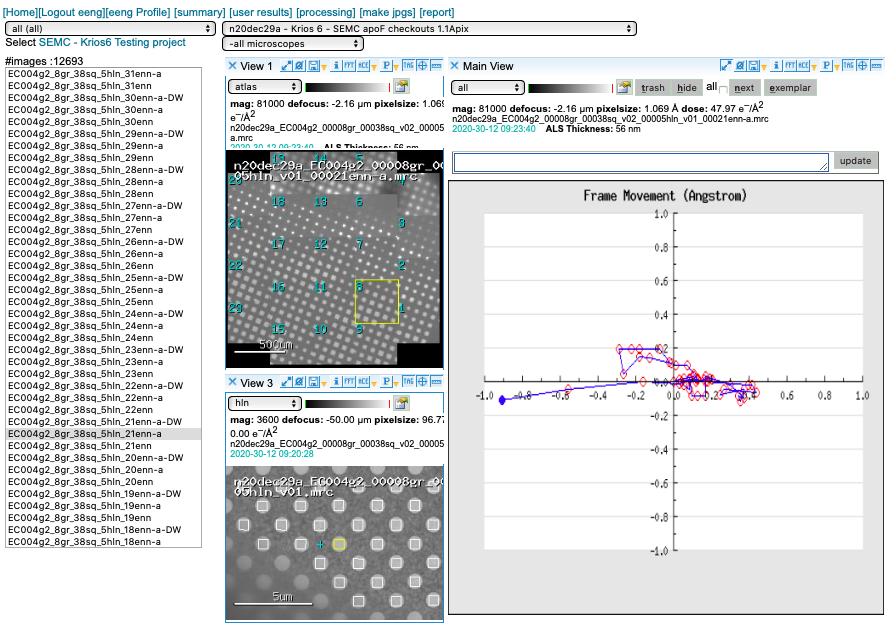
We run MotionCor2 using 5×5 patches. Bin1 for counting and Bin2 for super-res. You may access this plot through the arrow icon.
CTF estimation
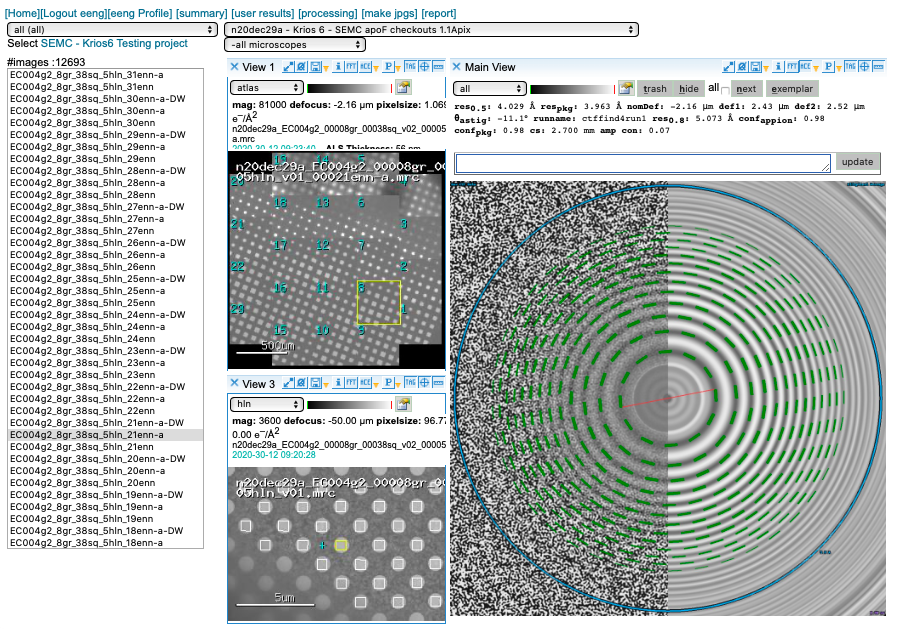
We prioritize our GPUs for motion correction and use CTFFind4 on our spare CPUs for CTF estimation. You may access this plot through the “ACE” icon.
Image tool
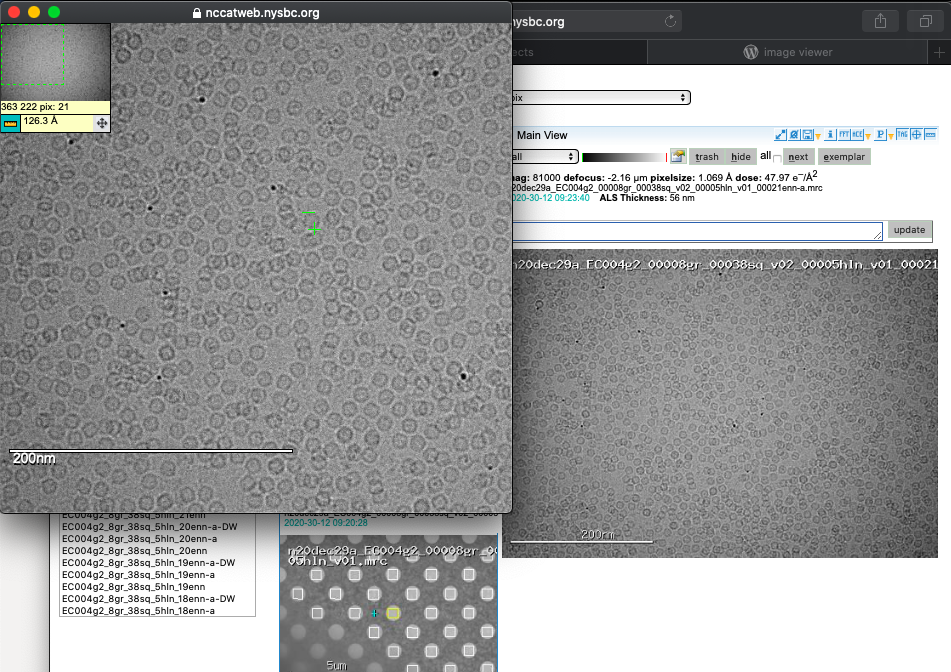
A common question is what size is my particle? By clicking on the image you can expand your view and engage a ruler tool to measure your macromolecules of interest.
Reports
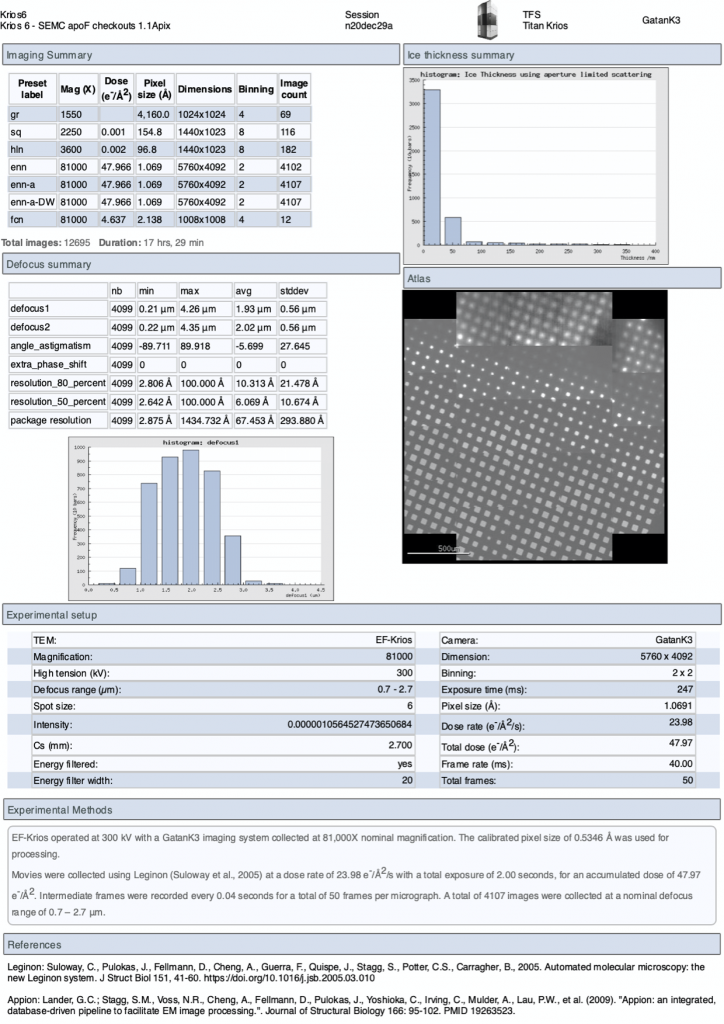
On the upper menu in the webpage you may also access a “Report” page to give you a summary of the experiment. This report updates in real-time.
What are all these presets?
- gr – images montaged together to create a whole grid Atlas
- sq – medium magnification image used to find squares
- hl – medium magnification image used to find holes
- enn or esn – high magnification image in counting or super res, respectively
The Leginon files names are explicit in case you lose web viewer functionality you can trackback to the parent images related to your current image.
Why are there 3 versions of enn?
Leginon aligned sum images are as follows:
- *enn.mrc – unaligned sums and may be 8×8 early return images
- *enn-a.mrc – aligned sums (by MotionCor2)
- *enn-a-DW.mrc – aligned dose weighted sums (by MotionCor2)
Additional tools in our web viewer include for each image the nominal defocus, estimated pixel size, total dose of the movie, timestamp when the image was taken, and ice thickness monitoring. If you are curious about our Appion pre-processing tools you may schedule a small group training session through the NCCAT User Office.
Why do my enn images look funny?
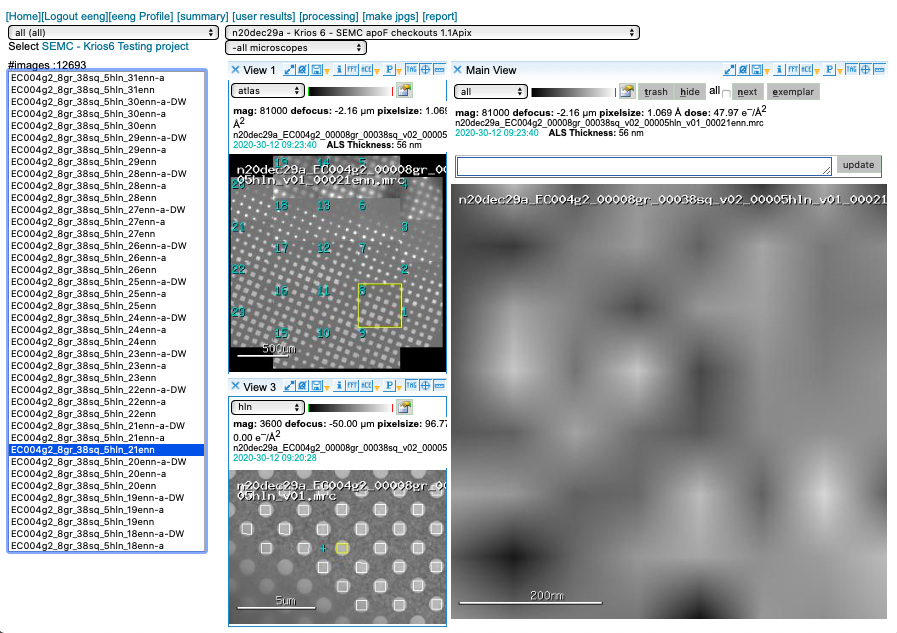
General frame-saving camera workflow in Leginon requires a 2D image array return from the camera to conclude its operation. To speed up data collection we return a Guassian-distributed image at 8×8 pixels. Read more [here].
After your session / getting your data
Please download your data via Globus. It may be easier to download the raw TIFF LZW or MRC data after a session is finished, but the Leginon aligned sum images (*enn-a-DW.mrc) are relatively small and that would be useful if you wanted to use them for initial processing feedback. We will store your data for 1 month, then it will be queued for deletion. If you have issues downloading your data please let us know.
Your data is only accessible through the spokesperson’s login located in a few places:
- the data on nccatweb.nysbc.org is located in /beegfs/leginon/[username]/ ,
- the direct detector data (movies in TIFF LZW from the Gatan K3 or EER from the TFS Falcon4i) is located in /beegfs/frames/[username] ,
- the pre-processing is in the Appion folder and may be regenerated through processing and located in /beegfs/appion/[username] , and
- the references are located in the folder called references in the frames directory. You may see a couple because we updated the gain after we started the session. So the latest one is the one to use.
Data directories
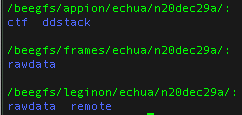
Note: At minimum users should always download all the data in the frames folder because this folder contains your raw data and all other files may be regenerated from your raw data.
References
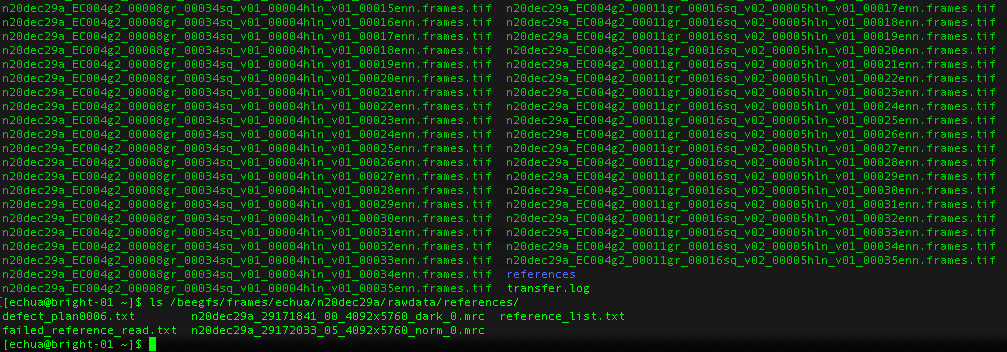
What about gain references?
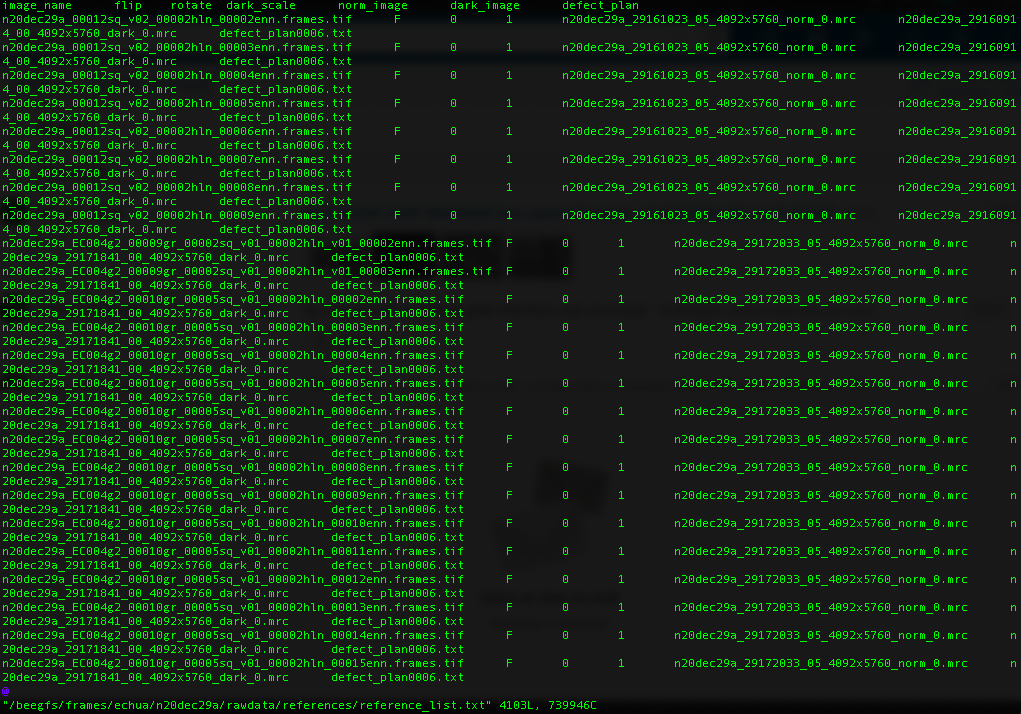
For Gatan K3 images gain correction requires a y-flip for most motion correction programs (e.g. cryoSPARC). The K3 is hardware dark corrected and only the gain reference is needed. The name of the gain reference would be in a format, such as “n20dec29a_29172033_05_4092x5760_norm_0.mrc”. If there are multiple references you should use the latest. If a new gain reference was taken in the middle of the session it would be annotated in the” reference_list.txt” file.
For TFS Falcon3 images, the MRC movies in your frames folder are already dark and bright corrected. No reference files are needed for processing. If collecting on a TFS Falcon4i system, then the images would be in EER format. In this case, the references would be found in your frames folder.
For more information read our wiki [here].
Reference list
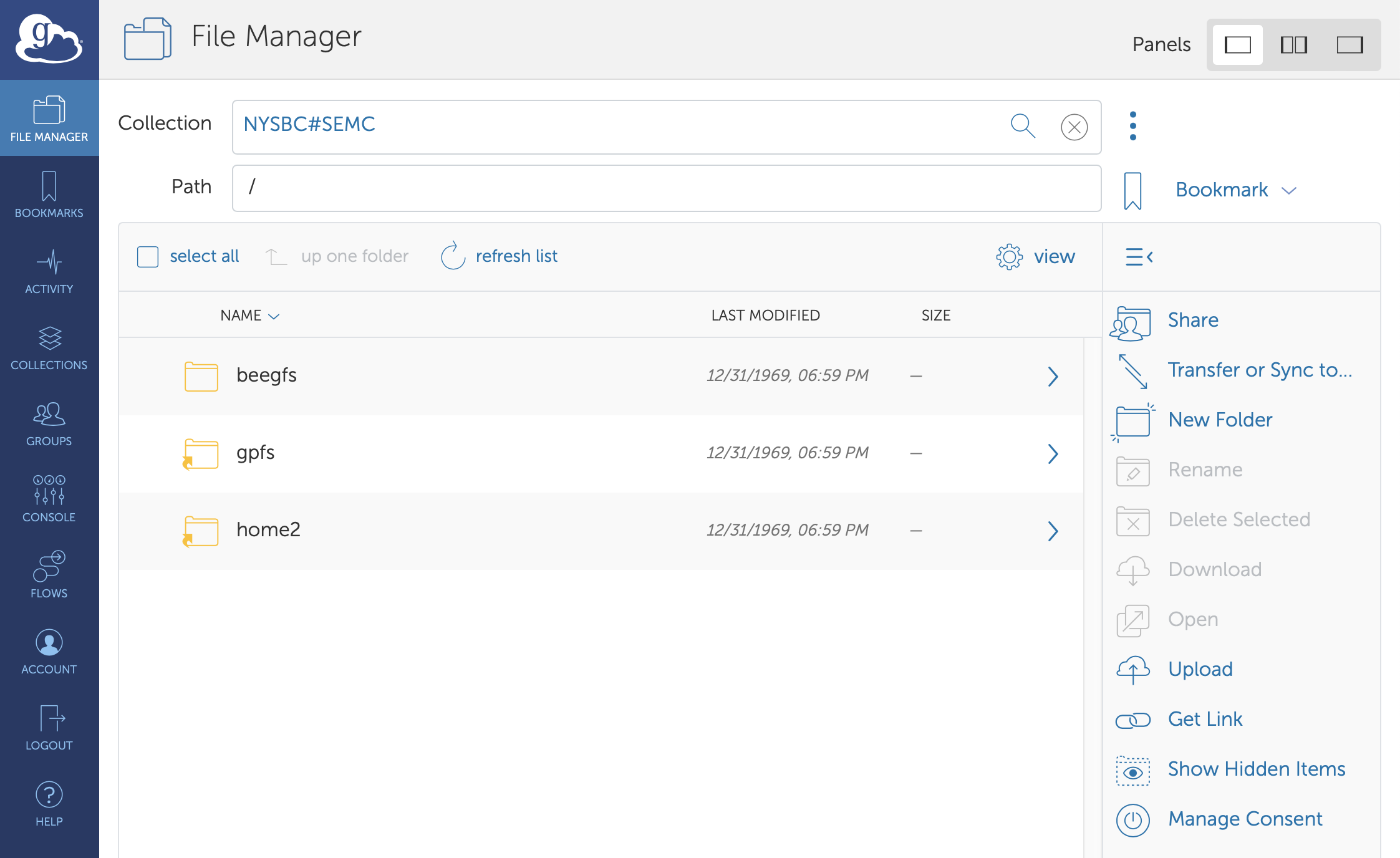
We use Globus, a non-profit service for secure, reliable research data management, for data transfer.
Please note that you are responsible for your data and samples. In general, direct detector data will be queued for deletion in a month. Your Leginon images are not backed up and may be converted to JPEG versions within 1 year. We will make our best efforts to preserve the functionality of the image viewer, but do not archive data. If you are having trouble with Globus let us know. If it is the endpoint we maintain our IT may assist, but Globus is a service we subscribe to and general help may also be found on their website: https://www.globus.org/
Globus file manager
At the end of your session
At the end of your session please make sure to contact the NCCAT User Office to work out any sample/grid logistics and fill out the end of session forms.
