Shipping Information
Labeled grid box user policy
Last updated: 05/05/2025
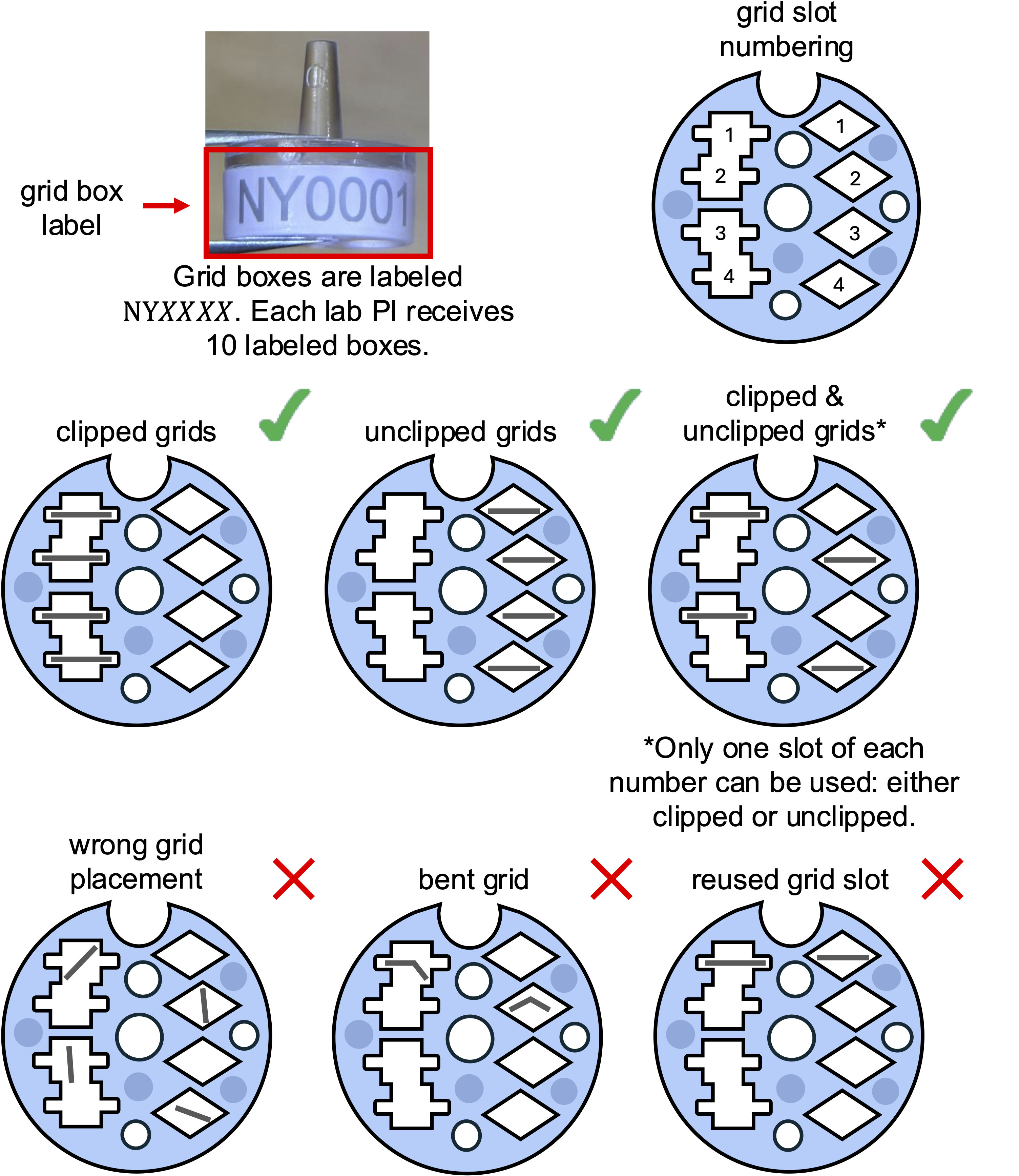
Starting April 2025, users must drop off samples in labeled grid boxes (e.g., NY0001, see image below) provided by NYSBC.
- Each lab PI will receive 10 labeled grid boxes for free.
- Grid boxes are not for long-term storage or regular lab use.
- Grid boxes are for tracking grids within NYSBC and improving communication between users and NYSBC staff.
If NYSBC staff accidentally break or lose a grid box, a free replacement will be provided.
If a lab PI needs more than 10 labeled grid boxes or a replacement, additional boxes can be purchased for $20 USD each. This purchase includes the labeled grid box, grid box cap, and shipping. Please note that the grid box and cap cannot be purchased separately. To place a purchase order, please email help@nysbc.org and NYSBC will provide an invoice to the lab PI.
Documentation:
- If you are shipping grids, fill out a shipping manifest and an operator form. If you are only shipping sample in tubes, you will only have to submit a shipping manifest. Both forms can be found at the following link: https://nccat.nysbc.org/information/
- Before shipping, email all forms to NCCAT User Office (nccatuseroffice@nysbc.org).
- Inside your dry shipper, include printed copies of the forms, along with a return shipping label.
- Notify the NCCAT User Office on the day the dry shipper is sent out.
Our shipping address is:
SEMC Shipping and Receiving Team
New York Structural Biology Center
89 Convent Ave
New York, NY 10027
Tel: (212) 939-0660
Shipping considerations:
- Try to avoid having your shipment arrive at our facility on weekends or national holidays. NYSBC will be closed and there will not be anyone onsite to receive your dry shipper.
- Delays in shipping are not uncommon, so make sure to properly charge the dewar as recommended in the manufacturer’s instructions, in order to maximize the static hold time. Securely tying down the dewar’s lid ① will also help prolong its static hold time.
- If dropping off grids instead of shipping, schedule a date and time with the NCCAT User Office (nccatuseroffice@nysbc.org). A shipping manifest and an operator form must be emailed to NCCAT User Office before drop-off.
Grid boxes:
- The tops of your grid boxes should be properly screwed down before shipping, as they can come loose in transit, which could result in the grids being lost ②. However, avoid fastening them too tightly, as expansion caused by the change in temperature can result in the lids becoming stuck.
- Label your grid boxes as per the instructions on the shipping agreement form. If the exact date of your session is unknown, use a more general one, such as “April, 2021”, or “Spring, 2021


If using falcon tubes instead of a puck system:
- Drill a hole in either the cap or side of the tube to allow LN2 to pour in and N2 to vent out. LN2 will expand 696 times as it vaporizes, which can cause a falcon tube to crack or explode ③ when unable to vent out.
- Securely fasten the cap of the tube. If it comes off during shipping the grid boxes can get knocked around the dewar ④.
- Please use frozen kimwipes or aluminum foil to pack inside the 50 mL conical tube with your grid boxes to prevent jostling during shipment.



GUP2 SAMPLE SHIPPING
Make sure your sample meets these criteria:
- Concentration: at least 2X higher than your current plunging conditions
- Glycerol: 0–10 %
- Detergent: none to as low as possible
- Stability: at least 1 hour at RT
- Volume: at least 20 µl
GUP2 Sample technical details.
Provide the following information about your sample:
- Name of macromolecule/complex
- Molecular weight and symmetry
- Storage buffer
- Ligands/binding partners in sample
- Sample storage conditions
- Current plunge freezing protocol including:
• Grid type
• Glow discharge protocol
• Sample concentration
• Pre-plunge preparation protocol (e.g. incubation, adding ligands etc.)
• Plunge freezing protocol
